

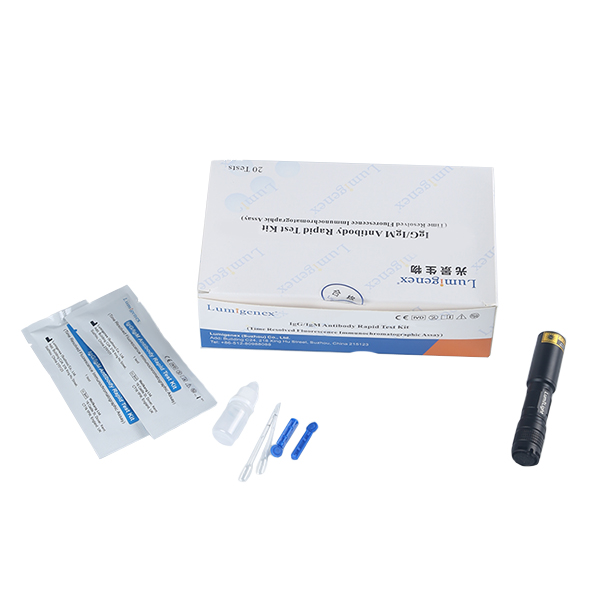

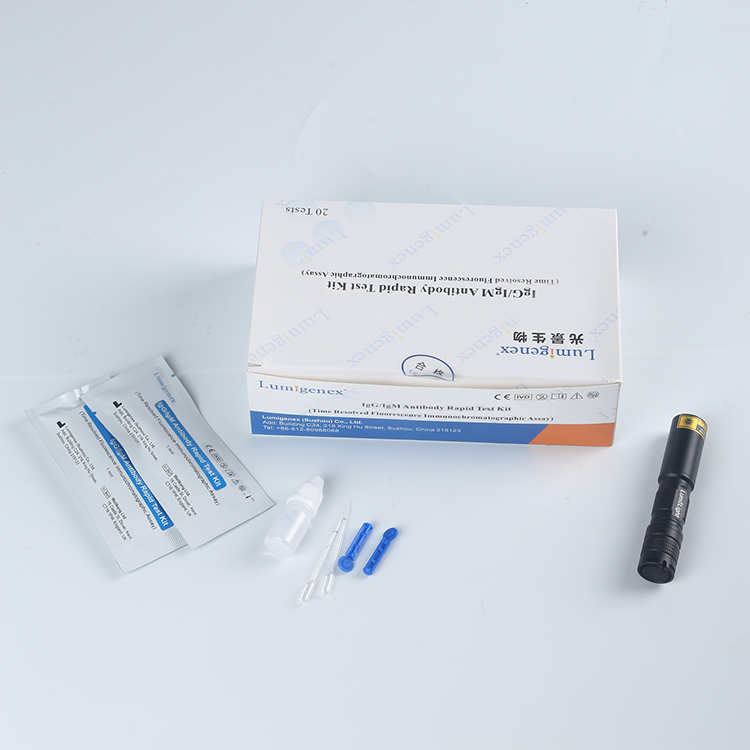
FOB Price : Get a Price/Quote
Min.Order : 1000 Piece(s)
Certification : ISO13485,CE
Brand Name : Lumigenex
Payment Terms : T/T
brand name : Lumigenex
certification : ISO13485,CE
min.order : 1000 Piece(s)
warranty : meet CE requirement
payment terms : T/T
Packaging : 20 Tests/Kit
Specification : 20 Tests/Kit
place of origin : China

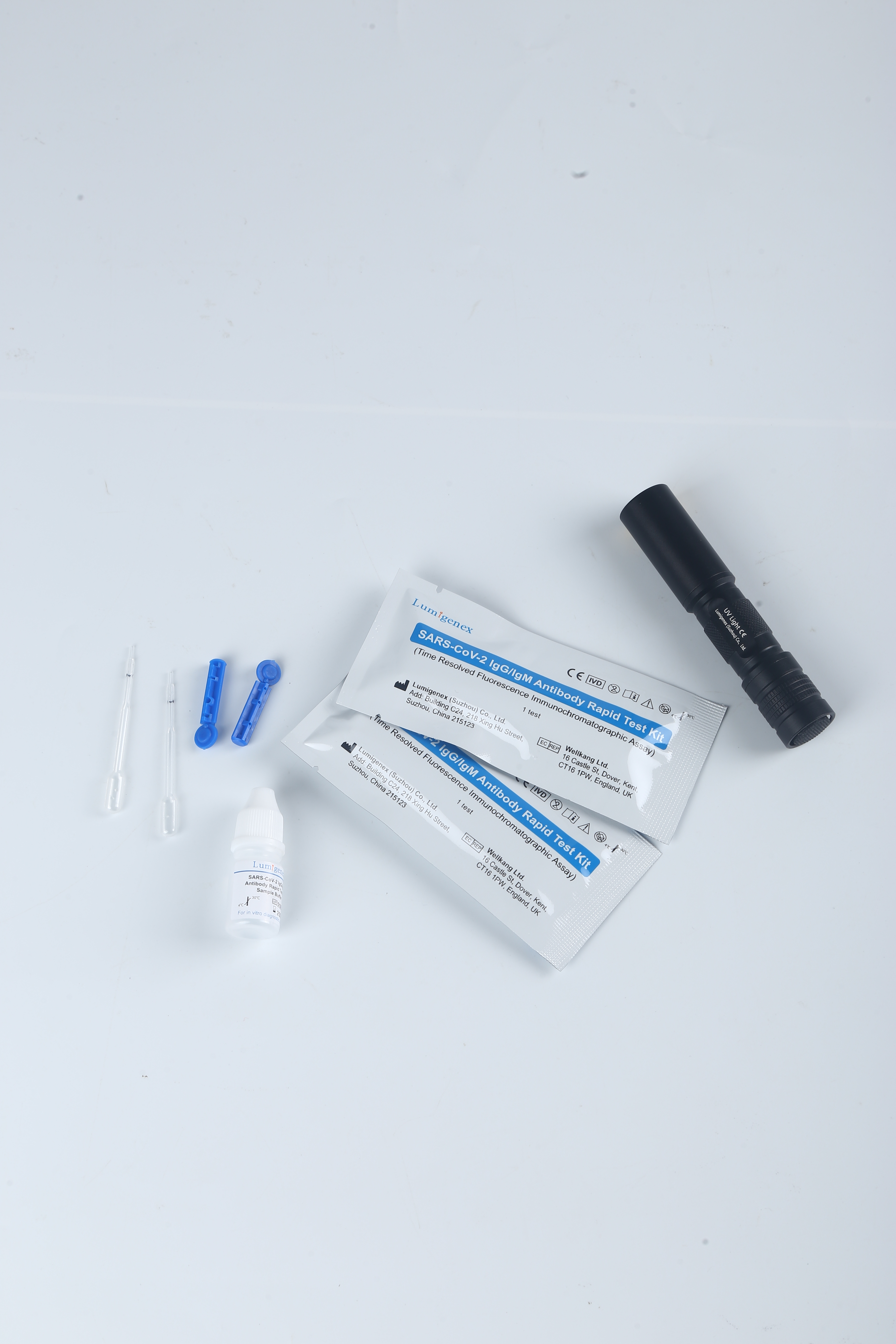
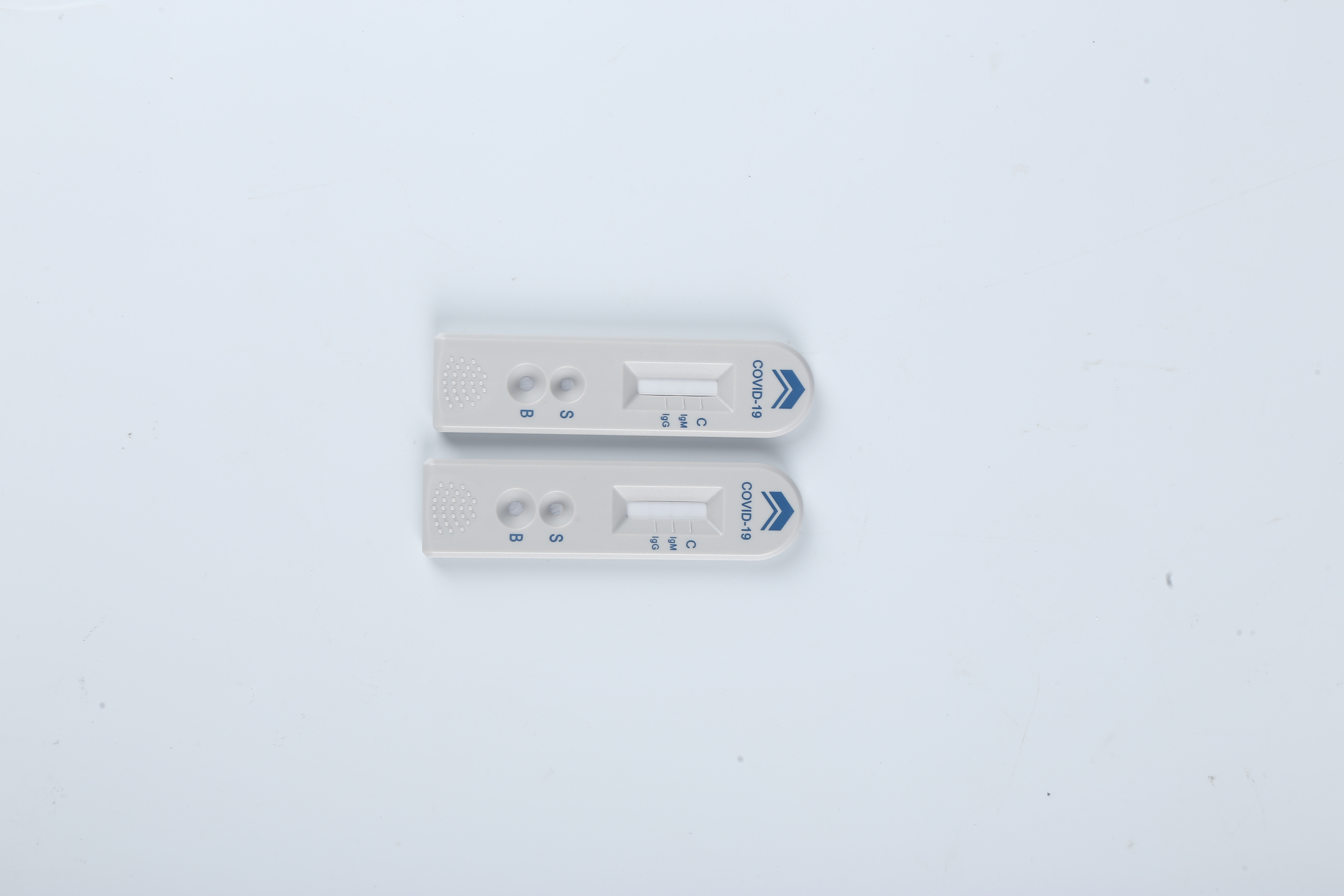
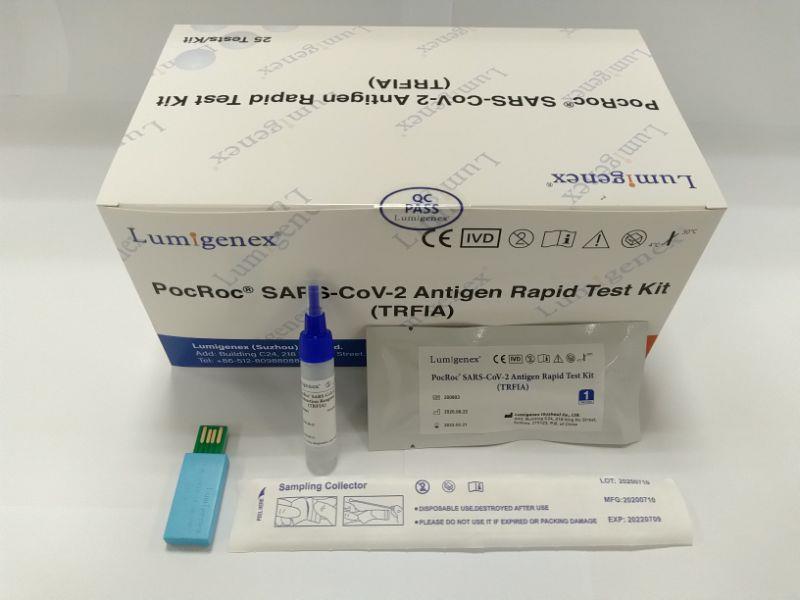
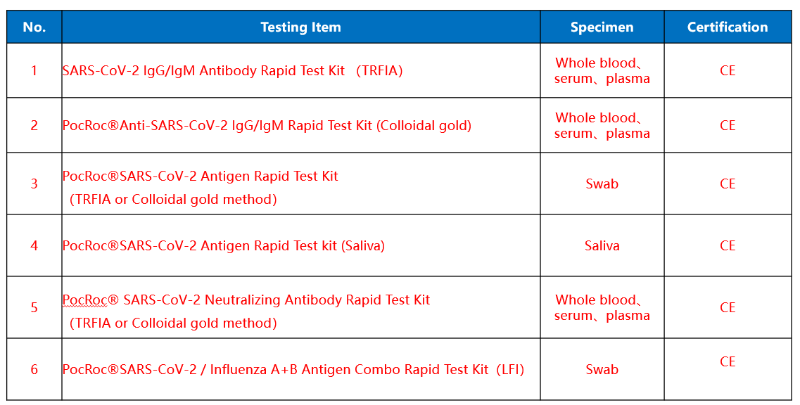
This rapid test kit is intended for in vitro qualitative detection and differentiation of antibodies to Severe Acute Respiratory Syndrome Coronavirus 2 in serum/plasma/whole blood samples specimens from patients suspected of Coronavirus Disease 2019 infection. It is used as an aid in diagnosing symptomatic or symptomatic individuals of acute infection together with molecular tests or clinical information. They can also identify individuals who have previously been infected with SARS-CoV-2 virus and recovered.
The test has been authorized only for detection of IgM and IgG antibodies specific to SARS-CoV-2, not for any other viruses or pathogens, and results should not be used as the sole basis to diagnose or exclude SARS-CoV-2 infection or to inform status.
PACKING SPECIFICATION
20 test strips/kit
Sample Buffer (mL*bottle): 3.0*1
REAGENTS AND MATERIALS
Each strip is sealed in a foil pouch with two items inside: one testing strip and one desicant.
Sample buffer:0.9% NaCl.
Disposable micropipettes.
Instructions for use.
MATERIALS REQUIRED BUT NOT PROVIDED
1. Timer
2. 75% alcohol wet wipes(order separately)
3. Disposable Blood Lancet(order separately)
4. A UV flashlight (order separately)
5. TRFIA analyzer/IC card (order separately)
TECHNOLOGY ADVANTAGES
Globally exclusive Time-Resolved Fluorescence Immunochromatography patented technology: the most accurate and sensitive POCT technology.
·Nanoscale phosphor (fluorescence) optical microsphere probe technology (Lumigenex proprietary invention patent)
·TRFIA Technology (Kimberly-Clark worldwide exclusive license)



 Ordinary
Ordinary
 verified
verified
Business Type Service
Country / Region Jiangsu,China
Main Products POCT Diagnostic Reagents, Diagnostic Tests
Main Markets SouthAmerica,EasternEurope,SoutheastAsia,Africa,MidEast,WesternEurope
Brand : Lumigenex, PocRoc
Min.Order : 1000 Piece(s)

Brand : Lumigenex
Min.Order : 1000 Other(s)
Brand : Lumigenex
Min.Order : 1000 Piece(s)
Brand : Lumigenex
Min.Order : 1000 Piece(s)
Brand : Lumigenex
Min.Order : 1000 Piece(s)
brand name : Lumigenex
certification :
fob price :
min.order : 1000 Piece(s)
warranty : meet CE requirement
payment terms : T/T
Packaing : 20 Tests/Kit
Specification : 20 Tests/Kit
Trademark : Lumigenex
Production Capacity :
place of origin : China
Manag Certifica : ISO13485,CE



This rapid test kit is intended for in vitro qualitative detection and differentiation of antibodies to Severe Acute Respiratory Syndrome Coronavirus 2 in serum/plasma/whole blood samples specimens from patients suspected of Coronavirus Disease 2019 infection. It is used as an aid in diagnosing symptomatic or symptomatic individuals of acute infection together with molecular tests or clinical information. They can also identify individuals who have previously been infected with SARS-CoV-2 virus and recovered.
The test has been authorized only for detection of IgM and IgG antibodies specific to SARS-CoV-2, not for any other viruses or pathogens, and results should not be used as the sole basis to diagnose or exclude SARS-CoV-2 infection or to inform status.
PACKING SPECIFICATION
20 test strips/kit
Sample Buffer (mL*bottle): 3.0*1
REAGENTS AND MATERIALS
Each strip is sealed in a foil pouch with two items inside: one testing strip and one desicant.
Sample buffer:0.9% NaCl.
Disposable micropipettes.
Instructions for use.
MATERIALS REQUIRED BUT NOT PROVIDED
1. Timer
2. 75% alcohol wet wipes(order separately)
3. Disposable Blood Lancet(order separately)
4. A UV flashlight (order separately)
5. TRFIA analyzer/IC card (order separately)
TECHNOLOGY ADVANTAGES
Globally exclusive Time-Resolved Fluorescence Immunochromatography patented technology: the most accurate and sensitive POCT technology.
·Nanoscale phosphor (fluorescence) optical microsphere probe technology (Lumigenex proprietary invention patent)
·TRFIA Technology (Kimberly-Clark worldwide exclusive license)

