trending topics
market reports
-

Registration Now Open: MEDICAL JAPAN 2026 OSAKA – Western Japan’s Largest Healthcare Trade Show
2026-02-10
-

MEDICAL JAPAN 2025 OSAKA Returns to Showcase Global Innovations
2025-02-17
-

Visit MEDICAL JAPAN 2023 TOKYO and take full advantage of the business opportunities!
2023-09-01
-

US to distribute 400 million free N95 masks at CVS, Walgreens in COVID fight
2022-01-21
-

Ethiopia receives additional 2.2 mln doses of Chinese-donated COVID-19 vaccines
2022-01-21
-
Hong Kong researchers say they develop novel material able to kill COVID-19 virus
2022-01-14
-

10 million more Chinese doses on way for Kenya
2022-01-14
-

Sino-African ties on track for a brighter future
2022-01-07
-
Efforts urged to boost COVID-19 vaccine production capacity in poor countries
2022-01-07
-
UAE approves Sinopharm's new protein-based COVID-19 vaccine
2022-01-07
Chinese inactivated COVID-19 vaccine starts phase-3 clinical trials in Argentina
2020-08-24
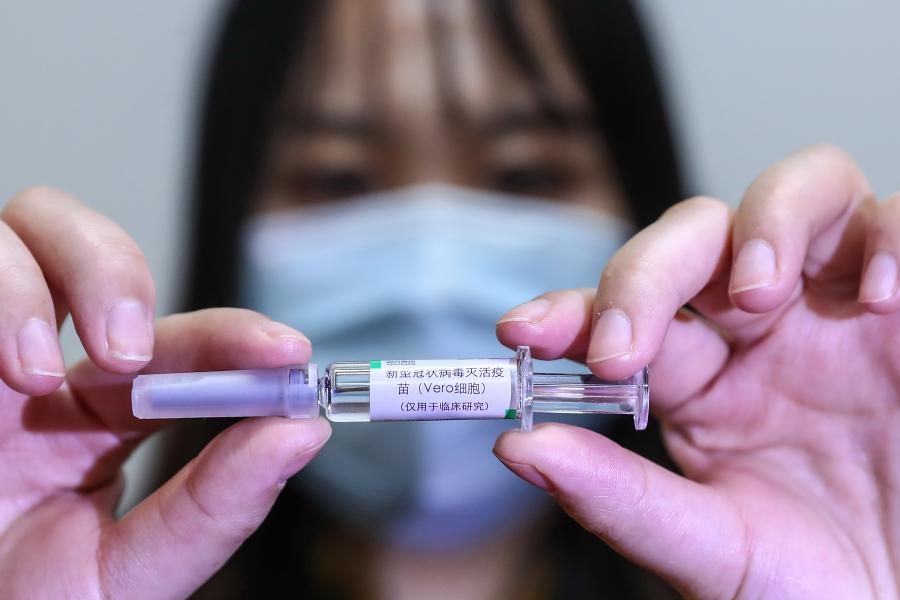
A staff member displays a sample of the COVID-19 inactivated vaccine at a vaccine production plant of China National Pharmaceutical Group Co., Ltd. (Sinopharm) in Beijing, capital of China, April 10, 2020. (Xinhua/Zhang Yuwei)
BEIJING, Aug. 22 (Xinhua) -- An inactivated COVID-19 vaccine developed by China has started phase-3 clinical trials in Argentina, according to its developer China National Biotec Group (CNBG), which is affiliated to Sinopharm.
The launch ceremony of the phase-3 clinical trials was held in Beijing Friday after the CNBG obtained the certificate of approval for the process.
This is an achievement brought about by CNBG's international cooperation in a bid to develop a COVID-19 vaccine. The inactivated vaccine received approval for phase-3 clinical trials in the United Arab Emirates on June 23, and in Peru and Morocco on Thursday.
The CNBG will work together with a company in Argentina to promote research and development related to the inactivated vaccine.
Liu Jingzhen, chairman of Sinopharm, said the phase-3 clinical trials will help fight the epidemic and contribute to the building of a community of health for all.
(People's Daily)



 My Member
My Member Message Center
Message Center











